Search
Emerging power sources of Electric Vehicles: Part 1
- TEAMXLR8

- Oct 12, 2021
- 15 min read
Updated: Jan 28, 2022
In the light of world shifting to green economy, the responsibility of replacing the petrol and diesel sources with some better and greener sources of fuel is really a big deal and that to, for a densely populated country like INDIA it seems like easier said than done.
For entrepreneurs and researchers, it is important to understand the mindset and the problems of mass, and then to work on the problems and build up a problem-solving product. Let’s see the following story to understand some basic but worthy problems of people-

One fine morning Ravi wakes up an and gets an urgent call to immediately attend an important meeting in his office. Ravi has both an electric car and a petrol car, but the problem is that neither the electric car is charged nor the petrol car is filled with fuel .In such instances instead of charging the electric car for around average of 2 hrs. , he would definitely fill the normal car with fuel which would hardly take seconds instead of waiting for the battery to be charged.
Similarly, if we keep a fully charged electric car idle for like 3 to 4 weeks, we will see that there will be a significant amount of loss of charge, but for a full tanked petrol car there will be less loss (due to evaporation and other factors) compared to an electric car.

People cannot go with an electric car for long distance. Always there will be a range anxiety as well as due to lack of charging infrastructure a normal middleclass man will not prefer an electric car instead, he would definitely go for a petrol car after all everyone wants to save time. In short Limited driving range, high costs, battery issues, and a spotty charging infrastructure are the main challenges for battery electric vehicles (BEVs). Although this is eco-friendly but its challenges hide its main importance and need in this greatly polluting world. Over the past few years, researchers from all over the world are trying to build a fast charging long battery life (more cycles). Who knows may be in the near future we can see great battery technologies that could charge up in seconds with high energy and power density i.e. capable enough to hold charge for long time and as well as long lifespan.
Graphene:
The line which we draw on our paper with pen or pencil is itself has certain thickness but still it could not be termed as 2D, but definitely this particular thing (GRAPHENE) IS 2D. Yes, Graphene has only single layer of atoms of carbons linked with each other in hexagonal structures. Sometimes it is called as single layer of graphite.
Over the past few years, we have seen a huge and rapid development of battery technologies especially lithium ion batteries and its variations with different other substances. We have seen great companies like TESLA MOTORS, SAMSUNG to provide batteries with fast charging and even new innovative Bluetooth charging facilities. But the problem remained unsolved i.e. no such technologies couldn’t be created that can charge up in seconds.

Discovery of Graphene:

In 2004, two scientists, Andrew Giem and Konstantin Novoselov at the University of Manchester, created the first sample of graphene. Andre and Kostya frequently held 'Friday night experiments' - sessions where they would try out experimental science that wasn’t necessarily linked to their day jobs.
One Friday, the two scientists removed some flakes from a lump of bulk graphite with sticky tape. They noticed some flakes were thinner than others. By separating the graphite fragments repeatedly, they managed to create flakes that were just one atom thick. Their experiment had led to graphene being isolated for the very first time.
Advantages of graphene batteries over lithium ion batteries:
Smaller, slimmer battery, graphene is lightweight. It’s when you stack 3 million layers of graphene is that you get 1 mm thickness. That should be enough to tell you that graphene batteries aren’t going to take much space in your future smartphone. It will allow manufactures to place higher capacity batteries in your phones, tablets, laptops, and more. Higher capacity: Graphene has a higher energy density as compared to lithium-ion batteries. Where the latter is known to store up to 180 Wh per kilogram, graphene’s capable of storing up to 1,000 Wh per kilogram. So, you can have a higher capacity graphene battery pack of the same size as the lithium-ion battery.
Other benefits include faster charging times, thermal management and greater safety. Graphene batteries are definitely the next big thing because carbon is in abundance as compared to Lithium, which is a rare metal.”
The rarity of lithium is another reason why comparing the graphene battery with current lithium makes commercial sense.
Graphene-enhanced Battery products moving towards Commercialization:
Graphene-based batteries have exciting potential and while they are not yet fully commercially available yet, R&D is intensive and will hopefully yield results in the future.

i) In December 2018, India-based Log 9 Materials announced that it is working on graphene-based metal-air batteries, that in theory may even lead to electric vehicles that run on water. The metal air batteries use a metal as anode, air (oxygen) as cathode and water as an electrolyte. A graphene rod is used in the air cathode of the batteries. Since Oxygen has to be used as the cathode, the cathode material has to be porous to let the air pass, a property in which graphene excels. According to Log 9 Materials, the graphene used in the electrode is able to increase the battery efficiency by five times at one-third the cost.
ii) In November 2017, Samsung developed a unique "graphene ball" that could make lithium- ion batteries last longer and charge faster. In fact, Samsung Advanced Institute of Technology (SAIT) said that using the new graphene ball material to make batteries will increase their capacity by 45% and make their charging speed five times faster. It was also said that the Samsung battery that will use this graphene ball material will be able to maintain a temperature of 60 degrees Celsius that is required for use in electric cars.
iii) In November 2016, Huawei unveiled a new graphene-enhanced Li-Ion battery that can remain functional at higher temperature (60° degrees as opposed to the existing 50° limit) and offers a longer operation time - double than what can be achieved with previous batteries. To achieve this breakthrough, Huawei incorporated several new technologies - including an anti-decomposition additive in the electrolyte, chemically stabilized single crystal cathodes - and graphene to facilitate heat dissipation. Huawei says that the graphene reduces the battery's operating temperature by 5 degrees.
iv) In June 2014, US based Vorbeck Materials announced the Vor-Power strap, a lightweight flexible power source that can be attached to any existing bag strap to enable a mobile charging station (via 2 USB and one micro USB ports). the product weighs 450 grams, provides 7,200 mAh and is probably the world’s first graphene-enhanced battery.
Many other companies are also working on incorporating graphene into various kinds of batteries. However, this too less information for graphene-based technologies compared to its evolution and developments that is covered in this.
NDB: Nano Diamond Battery:

IMAGINE A SMART PHONE DOESN’T REQUIRE CHARGING
Uninterrupted device usage.
Higher computational power.
Possibility of a new generation quantum smartphones.
IMAGINE NO REFUELING STATIONS REQUIRED
Uninterrupted drive mode.
Sustainable energy.
Higher mileage.
IMAGINE A MINIATURE POWER GENERATOR FOR EVERY DEVICE!
Cost effective to counter declining sales.
Power outlet independent devices.
Higher device computation power.
No power heating issue.
Personal quantum computers.
IMAGINE ELECTRICITY FOR EVERYONE & EVERYWHERE
Schools, colleges, buildings
Envisioning a world where remote and disadvantaged populations can easily access electricity
To reinvent electricity & create a planet without fossil fuels by 2040

US start up NDB, a company that says it has created the first and only universal, self-charging nano-diamond battery that provides thousands of years of charge, announced that two of its proofs of concept achieved a breakthrough 40% charge.
Some Interesting Facts:
The California-based start-up NDB has unveiled a battery that uses nuclear waste and lasts up to 28,000 years.
The power of the nano-diamond battery comes from radioactive isotopes used in nuclear reactors.
Its radioactive core is protected by multiple layers of synthetic diamonds, one of the hardest materials to damage or break.
The energy is absorbed in the diamond through inelastic scattering, which is used to generate electricity.
The battery can be used to power devices and machines of any size, from aircraft and rockets to electric vehicles and smartphones.
Nima Golsharifi, CEO and Co-Founder of NDB, said: “As members of society, we are extremely concerned about the welfare of the planet and are focused on lowering climate change to protect our planet for future generations.
“With the NDB battery, we have achieved a massive, ground-breaking, proprietary technological breakthrough of a battery that is emission-free, lasts thousands of years and only requires access to natural air in order to power devices.”
The company says the development of the first commercial prototype battery is currently underway and will be available later this year.
How does NDB works?

The main concept is to reuse the radioactive waste which when undergoes decay of highly energetic particles to produce electric current.
14C undergoes beta decay, in which it emits a low-energy beta particle to become Nitrogen-14, which is stable (not radioactive).
(14:6)C→ (14:7)N+ (0:−1)β
It also lasts long.
Proposed Manufacturing:
In graphite-moderated reactors, fissile uranium rods are placed inside graphite blocks. These blocks act as a neutron moderator whose purpose is to slow down fast-moving neutrons so that nuclear chain reactions can occur with thermal neutrons.[9] During their use, some of the non-radioactive carbon-12 and carbon-13 isotopes in graphite get converted into radioactive 14C by capturing neutrons.[10] Once the graphite blocks are removed during station decommissioning their induced radioactivity qualifies them as low-level waste requiring safe disposal.

Researchers at the University of Bristol demonstrated that a large amount of the radioactive 14C was concentrated on the inner walls of the graphite blocks. Due to this, they propose that much of it can be effectively removed from the blocks. This can be done by heating them to the sublimation point of 3915 K which will release the carbon in gaseous form. After this blocks will be less radioactive and possibly easier to dispose of with most of the radioactive 14C having being extracted.
Those researchers propose that this 14C gas could be collected and used to produce man-made diamonds by a process known as chemical vapor deposition using low pressure and elevated temperature, noting that this diamond would be a thin sheet and not of the stereotypical diamond cut. The resulting diamond made of radioactive 14C would still produce beta radiation which researchers claim would allow it to be used as a beta voltaic source. Researchers also claim this diamond would be sandwiched between non-radioactive man-made diamonds made from 12C which would block radiation from the source and would also be used for energy conversion as a diamond semiconductor instead of convention silicon semiconductors.
Applications:

Automotive:
NDB can be used to further the electric vehicle revolution, pushing the industry into never seen before indicators of efficiency and longevity. NDB is ideally positioned to contribute and lead change in this industry by addressing the primary bottleneck in EV vehicles, the battery, one that can last as long as the vehicle’s does.
Aerospace:
The human desire to explore space is fueled by the excitement of exploring the unknown. Recent advances in space technology and the rise of the first manned electric aircrafts have led to increasing demand on their battery systems, hindered by concerns regarding longevity and safety. Satellites and space vehicles rely heavily on solar power which can be disrupted by hash space environment. NDB can be utilized to power drones, electric aircrafts, space rovers and stations whilst allowing for longer activity.
Consumer Electronics:
With NDB, every device you own, be it a smartphone or a laptop, can contain a miniature power generator, thus negating the need for constant charging, all whilst delivering a cost-effective solution to countering declining sales for manufacturers. Benefits include power outlet independent devices, increased device computation power and even the opportunity for the rise of personal quantum computing.
Medical Technology:
In situ medical devices and implantable such as hearing aids and pacemakers respectively can benefit from long battery life in a smaller package with added benefit of safety and longevity.
Industrial:
NDB’s safety, power output, and universality transforms this technology into an ideal industrial partner to bring power to many routine applications and those that are difficult to implement. Data centers, remote locations, and hostile environment applications of NDB make it an outstanding promise for productivity and futuristic applications.
Defense:
Defense related electronics and surveillance systems can benefit from NDB’s universal applicability and sustained power output, where cutting edge technology in line with proficient defense strategy can alleviate the mobile power concerns of this sector.
Water Based Batteries:
This is a very interesting technology and at the same time, quite a controversial one.
Principle:
There is energy in water which is chemically locked up in the atomic bonds between the hydrogen and oxygen atoms. This energy can be used to drive the vehicles.
Work done:
A Japanese company called Genepax unveiled their water powered car in 2008 in Osaka, Japan. The source of the water doesn’t matter here, any of the sources would work fine. An energy generator splits the water molecules apart into the constituent elements which are later used to power the vehicle. They use a membrane electrode assembly (MEA) to split the Hydrogen from the Oxygen through a chemical reaction. The cell needs only water and air, eliminating the need for a hydrogen reformer and high pressure hydrogen tank. Approximately a year after revealing their device, the company shut down citing lack of monetary funds to be the reason.
Stanley Meyer is another one who invented a water powered car using the process of electrolysis. He regulated the speed at which the oxygen unites with the hydrogen after entering the engine by using the non-combustible gases coming from the exhaust. Although he was successful in developing the car, several claims were made on his claims to be fraudulent. Most controversial of them being that he claimed that the heat released was greater than the electrical energy used, which seems to be quite impractical as this is a clear violation of First and Second laws of thermodynamics.


Working:
The key is to take electricity from the car's electrical system to electrolyze water into a gaseous mixture of hydrogen and oxygen, often referred to as Brown's Gas or HHO or oxyhydrogen. Typically, the mixture is in a ratio of 2:1 hydrogen atom to oxygen atoms. This is then immediately piped into the intake manifold to replace some of the expensive gasoline you've been paying through the nose for these last couple of months.

Water makes the perfect fuel source. It’s comprised of two hydrogen atoms and one atom of oxygen. When the water molecule is electrolyzed and separated into its two component atoms and oxidized as fuel, the result is equivalent to an energy output that is 2.5 times more powerful than gasoline. The by-product of the combustion is water vapor, totally pollution free, returning water back into the atmosphere.
There are some projects underway which use graphene rod along the metal plate and thus generate electricity with water as the base for the chemical reaction.
The Huge energy requirement to kickstart the process of electrolysis is the biggest challenge. This idea if successfully commercialized will increase the fuel economy and decrease the emissions, bills and dependence on foreign petroleum by anywhere from 15 to 300 percent. Further, Tata Motors is also funding a project on this Technology at M.I.T.
Sand-Based Batteries:
Silicon is the smartest element on earth and has the potential to do wonders. This technology is just an example where this amazing element is being used to harness energy and drive vehicles
Sand (SiO2) battery gives three times more battery life than the batteries we are currently using.
In these batteries, silicon is used in lithium-ion battery to achieve three times better performance than the current graphite li-ion batteries. The battery is still lithium-ion but it uses silicon instead of graphite in the anodes.
Scientists at the University of California Riverside have been focused on nano silicon for a while, but it's been degrading too quickly and is tough to produce in large quantities. By using sand, it can be purified, powdered then ground with salt and magnesium before being heated to remove oxygen resulting in pure silicon. This is porous and three-dimensional which helps in performance and, potentially, the life-span of the batteries. We originally picked up on this research in 2014 and now it's coming to fruition.
Battery tech start-ups like Silanano are bringing this technique to market and has seen big investment from companies like Daimler and BMW. They also claim that this solution can be put into use into the existing lithium-ion battery manufacturing, so it's set for scalable deployment, promising 20 per cent battery performance boost now, or 40 per cent in the near future.

Graphite Dual Ion Batteries:
Rechargeable graphite dual‐ion batteries (GDIBs) have attracted the attention of electrochemists and material scientists in recent years due to their low cost and high‐performance metrics, such as high power density (≈3–175 kW kg−1), energy efficiency (≈80–90%), long cycling life, and high energy density (up to 200 Wh kg−1), suited for grid‐level stationary storage of electricity. The key feature of GDIBs is the exploitation of the reversible oxidation of the graphite network with concomitant and highly efficient intercalation/deintercalation of bulky anionic species between graphene layers.
GDIBs operate as an electrochemical energy storage system employing reversible intercalation of anion species into the graphite cathode upon charge (oxidation of the graphite network).

Concurrently, intercalation, adsorption, alloying, or metal electroplating reactions might take place on the anode side of GDIBs. The examples of electrolytes used in GDIBs include ClO4−, bis(trifluoromethanesulfonic)imide (TFSI−), bis(fluorosulfonyl)imide (FSI−), PF6−, and BF4− anion‐based salts of Li, Na, or K, dissolved in alkyl carbonates as solvents. Such polar solvents allow for high salt concentrations and a broad voltage window for operation. Contrary to a Li‐ion battery (LIB) using graphite as an anode, GDIBs exploit the reversible oxidation of the graphite structure, that is, its cathodic functionality.
At the cathode:
𝑥𝐴− + Graphite ↔ Graphite(𝐴−)𝑥 + 𝑥𝑒−
At the anode:
(1/6) Li+ + (1/6)e− + C ↔ Li1/6C (intercalation)
𝑥Li+ + 𝑥e− + C (hard carbon) ↔ C(hard carbon)Li𝑥 (adsorption/insertion)
(4.4)Li+ + (4.4)e− + Sn ↔ SnLi4.4 (alloying)
Li+ + e−↔Li (electroplating)
VRFBs: Vanadium Redox Flow Batteries:
The switch to renewable energy is fundamental not only for protecting our planet from harmful emissions but also maintaining the energy dependent societies that we live in. The adoption of the Electric Car over conventional IC engine vehicles will help to use the renewable resources to meet our energy demands.
In addition to the Affordability of the EVs, there are some more barriers in its adoption, like:
Charging points of Electric Cars are not as available as conventional gas stations.
Current Charge Time of EVs is also more than the time it takes to refill IC engine cars with fuel.
Long journeys would require more frequent power charges for EVs than to make gas refills.
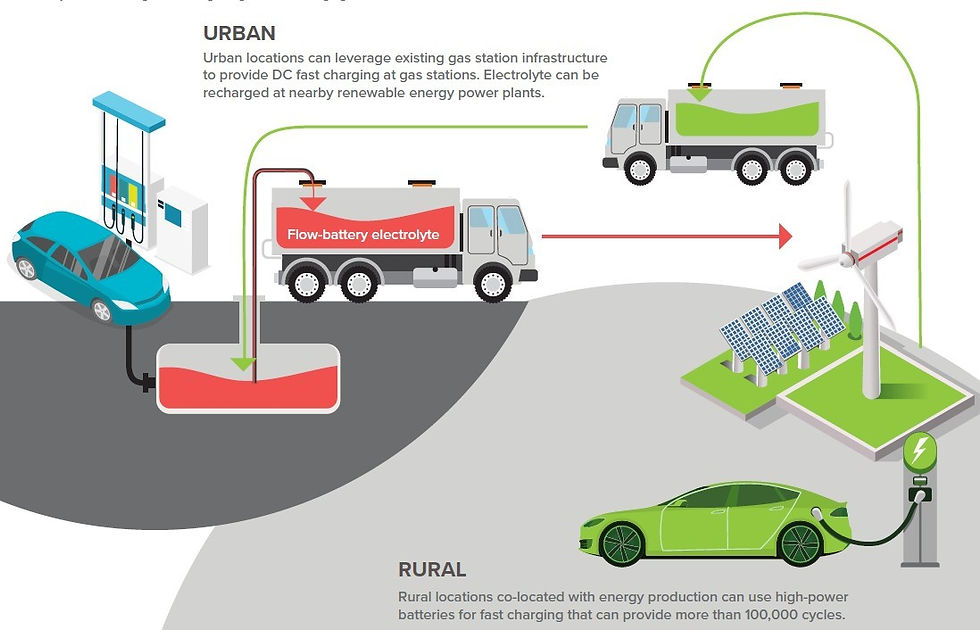
Flow Battery, A Game Changer:
Chemists based at Glasgow University have utilized a nano-molecule that has the capability of storing electric power or hydrogen gas in its new flow battery system. Instead of visiting charge points, cars could theoretically fill up at a gas station, as the system works on replacing fluid within the battery. An electric car owner would use the pumps to fill the battery with fresh electrolyte instead of the usual fossil fuel that is pumped into combustion engines, and the depleted electrolyte would be collected, recycled and reused by another customer. This means that no change to the infrastructure is required and current gas stations could be used to support electric vehicles, eliminating the concern over ease of use due to limited charge points.

As the energy is replaced through changing the electrolyte by pumping it directly into the system, recharge time is dramatically cut, meaning that filling up a regular car and recharging an electric one would take a similar time with the flow battery.
The fluid that releases power as both electricity or as hydrogen has the capacity to power the car for up to 3,000 miles, according to the latest tests. This has the impact of eliminating ‘range anxiety’ as a factor for not deciding to switch to electric. Tests have shown that with the new battery, drivers would be replacing the fluid with about the same frequency that they would refill a conventional car.
Advantages of Vanadium Redox Flow Batteries (VRFBs):

VRFBs have a lifespan of 20+ years.
VRFBs offer immediate energy release.
VRFBs are suitable for grid connection or off-grid settings– ideal for renewable energy.
VRFBs can discharge 100%, without any damage to the battery.
VRFBs are non-flammable.
They ensure power and energy can be scaled independently.
Vanadium electrolyte can be re-used and does not need to be disposed of.
The batteries can be cycled more than once per day.
They use only one element in electrolyte – V2O5.
VRFB energy storage guarantees uninterrupted power supply.
Working of VRFBs:

Redox flow batteries (red for reduction = electron absorption, ox for oxidation = electron release), are based on a liquid electrochemical storage medium. The positive and negative sides of a VRFB are separated by a membrane that selectively allows protons to go through. During charging, an applied voltage causes vanadium ions to each lose an electron on the positive side. The freed electrons flow through the outside circuit to the negative side, where they are stored. During discharging, the stored electrons are released, flowing back through the outside circuit to the positive side.
Challenges of Renewable Energy:
We also face challenges in order to use the Renewable Energies efficiently. Solar Energy provide solar power into the electricity grid, but all this electricity is coming through the grid at the wrong time of day. Between noon and 4 pm is a trough in power demand. It’s during peak hours of demand in the evening when all this excess energy can be utilized. Wind power encounters the same intermittency problems as solar power – sometimes the wind blows, sometimes it doesn’t, sometimes the sun shines sometimes it doesn’t.
Having the flexibility of being able to store electricity, and using it when the grid needs it, has always been one of the biggest challenges for renewable power.
Ideal for Renewable Energy:
Vanadium Flow Battery can store large amounts of energy almost indefinitely, which makes them perfect for wind/solar farms, industrial and utility-scale applications, to supply remote areas, or to provide backup power. With VRFB, we can put solar power and wind power into the battery, and we can put excess grid power into the battery at night.
Environmental Impact:
NanoFlowcell is a Swiss flow cell battery research and development company. Using NanoFlowcell technology can avoid the emission of 19,250 Megatons of CO2 every year from the combustion of fossil fuels in road traffic alone. That’s 75% of the CO2 Emission generated worldwide by transport and traffic.
Vanadium Redox Flow Batteries (VRFBs) Or Lithium-ion Batteries:
In Grid Energy Storage, Its No Contest. VRBs have many advantages over Li-ion Batteries as shown below:

Predicted Cost Analysis of Vanadium Redox Flow Batteries (VFRBs):




Comments